Brine freezing systems require attention to detail

Different brine freezing systems
Currently, two type of brine systems can be used to deep-freeze blocs of shrimp. The oldest method involves submerging stacks of shrimp blocks in a tank filled with chilled brine, and this is the immersion technique. In the early 2000s, a new method was developed by a French company at its shrimp processing facilities in Madagascar, and involving the spraying of chilled brine on the shrimp. This method, which today is used by several other companies, is called the shower technique.
For both methods, brine freezing is a two-phase process: a first phase to freeze the shrimp to a negative temperature, and then a second phase in a quick (roughly) 20-minute pass through a blast tunnel to reach a temperature lower than minus-18 degrees-C.

Immersion technique
The immersion technique, which consists of immersing the shrimp product to be frozen in chilled brine, is more efficient than the air blast freezer or than the contact plate freezer. However, it could still be improved, because the production is not continuous due to the need for the shrimp blocks to be grouped together in batches before being submerged in the brine; and because the flow of brine cannot really penetrate really the piles of blocks and come back out, so more time is needed for the cold brine to sufficiently penetrate the product.
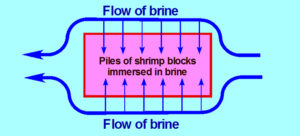
When the frozen shrimp blocks are removed from the brine (at temperatures of around minus-12 to minus-14 degrees-C), they must be rinsed quickly to remove the brine residues (at around minus-8 to minus-10 degrees-C) and then moved through an air blast tunnel to reach a core temperature of minus-18 degrees-C.
Shower technique
Developed to improve the original brine immersion freezing, the concept is to force the brine solution to completely go through and across the shrimp blocks. The chilled brine is applied and spread as a shower over the shrimp blocks that are moved on a conveyor belt. In this technique, with the brine going through the blocks, cold penetration is much faster and so is the freezing of the shrimp.

In addition to being faster, this method makes it possible to set up a continuous freezing process by placing the shrimp blocks on a moving conveyor belt that passes through the brine chamber and then into the blast freezer. There is no time wasted in the process from the entry of the fresh shrimp into the packing plant to the moment the product is frozen, which is very important to improve and maintain the quality of the final products.

Currently, there are two types of shower brine freezers. One uses an external plate heat exchanger and the other one has temperature exchange plates in the brine reservoir itself, as is also the case for the brine immersion technique.
The heat exchange capacity depends on the contact surface between the coolant and the brine. In the second case, it is therefore necessary to have a sufficiently deep brine tank/reservoir to be able to install the necessary heat exchange surface. This requires very large reservoirs and consequently a large volume of brine. This is not the case when using a plate exchanger located outside of the reservoir.
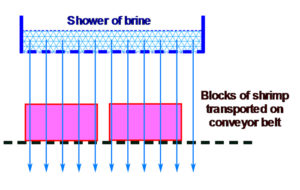
Fig. 2: Illustrated flow of brine and cold penetration in the case of showered brine.
Therefore, only about half of the brine volume is required with an external exchanger vs. placing the heat exchange plates in the reservoir. This is a very important aspect to consider because the less brine volume used, the easier and less expensive it is to manage and replace. We must also consider the eventual disposal of old brine solution, which is always cumbersome, and therefore the smaller the brine volume involved, the easier its disposal will be.
Preparation of new brine
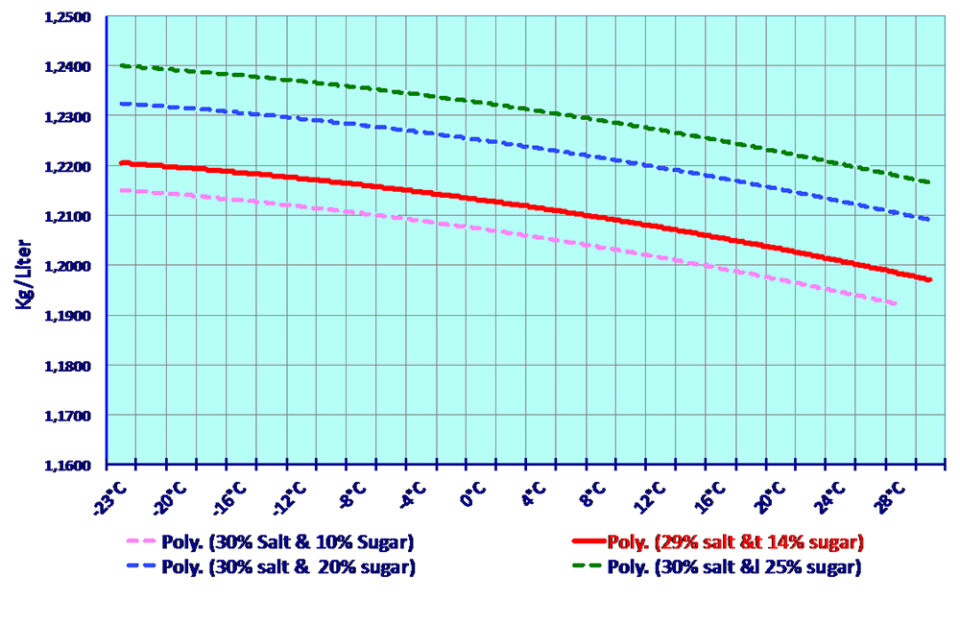
To prepare the brine, it is necessary to first dilute little by little all the salt needed by constantly checking the increase in density. When the density of 1.170 kg/Liter (or Kg/dm3) is reached with salt additions, gradual sugar additions begin. When all the sugar is diluted, the brine density should be around 1,200 kg/Liter (or Kg/dm3) at ambient temperature. The brine density can vary considerably depending on its temperature (Fig. 3), and must be closely watched and checked.
Brine management during the freezing process
During the process, the brine quality is diminished in two ways, by volume and by solute depletion. Consumption or depletion of brine volume can occur in three ways. By misting, which is the dissipation of the brine in the tunnel; by absorption by the shrimp (some brine is absorbed individually and other remains in internal shrimp spaces); and by lack of drainage, where the consumption of brine involves the volume of brine that remains trapped in the freezing molds. This depends and varies significantly based on the type, size and volume of the molds.
Depletion of solutes (salt and sugar) results from the penetration of salt molecules salt (NaCl) and sugar into the carapace and then into the flesh of the shrimp. This infiltration varies according to the size of the shrimp being processed, and is more important with smaller-sized animals. This brine volume depletion must be continuously compensated during the shrimp processing to prevent icing of the system, which can be accomplished in two ways. One, by compensating for the missing volume by adding brine having the original characteristics; and two measuring the quantities of solutes (salt and sugar) depleted in order to estimate how much solute supplementation needs to be added.
The brine concentration can only be controlled by checking the solution density, which must be not too high and not too low to prevent icing. If the density decreases, both salt and sugar must be added. However, just the density does not provide any information about the relative consumption/depletion of either or sugar, and so it is necessary to assess each at least once a day to properly adjust their concentration. For the salt concentration, this is done by a titration of chloride ions by the Mohr method, and the sugar concentration is estimated measuring the degrees Brix of the solution using a specific refractometer.
Salt or sugar must never be added as crystals (powder) directly to the brine reservoir of the freezer. If salt or sugar crystals were to be added directly to the brine tank, there is a risk that they would not dissolve and would sink to the bottom of the tank, forming a layer which is wasteful, useless and even harmful.
These additions must be done by returning the brine solution to a preparation tank where the necessary quantities of salt and sugar are added and dissolved, just like in the initial preparation of the brine. As the concentrations approach saturation, the salt and sugar dissolutions are more difficult, so it is necessary to assess the brine frequently and to avoid adding very large quantities at any one time.
Useful life of the brine
When the shrimp is frozen, the set point must be at least minus-18 degrees-C. Between processing events, the brine temperature can be allowed to rise to minus-8 degrees-C in order to prevent the propagation of any microorganisms present.
The brine can be used for several months. The main reason for replacing the brine is bacterial contamination, so it is important to monitor the evolution of brine contamination on a daily basis by bacteriological analyses; and to define acceptable levels and critical levels. When the critical level is reached, the brine must be replaced as soon as possible.

Boxing product in process
For many years, the shrimp were placed in plastic or stainless steel containers or molds before being frozen in the brine. After coming out of the final blast tunnel, it was necessary to remove the blocks of shrimp from the molds to transfer them into carton boxes. This was a delicate operation that could be even challenging for the workers who had to manually handle the frozen blocks. However, in recent years, manufacturers of cardboard boxes have developed solutions to make carboard boxes that are resistant to the brine shower. This simplifies the handling of the products at the exit of the freezer and their placement in master cartons before final storage in a cold room.
Perspectives
Brine freezing technology is an excellent approach to deep freeze shrimp. It provides the final product with an excellent texture, due to the fast freezing that allows for very high coefficients of heat transfer.
Compared with those of other traditional methods (blast freezing, plate freezing), this high coefficient also permits reducing significantly the amount of energy needed to freeze the products. This results in significant savings for the processor in terms of investment (less cold production equipment) and in terms of cost of production (less energy per kilo of product processed).
Brine freezing systems are not complicate but need special care during all the production process. Among the key recommendations: never add fresh water to chilled brine reservoir nor empty solid salt and/or sugar directly into the brine reservoir. The volume of the brine must be adjusted before to compensate for brine solutes depletion; and compensate for solutes depletion (both for volume and concentration) routinely during the process. Finally, always keep the brine at a temperature below minus-8 degrees-C.
Author
-

Hervé Lucien-Brun
Aquaculture & Qualite
9, rue Poupinel
F-78730 Saint Arnoult en Yvelines, France

