Common commercially utilized natural antioxidants are tocopherols and rosemary extracts

oxidative reactions.
Lipids are essential functional components in animal feeds and human foods. They provide a fundamental source of energy and essential nutrients, while also contributing to the palatability and texture of food. Preserving the quality of lipids is vital to retaining product shelf life and optimum animal production performance.
Lipids, lipid-containing ingredients, and feed primarily degrade through oxidation reactions that result in rancidity. Rancidity is typically associated with loss of nutritional value and the development of off odors. The oxidation of lipids can also result in lower feed conversion, decreased weight gain, and increased disease or mortality in culture animals. Lower energy value, protein quality, and vitamin retention in feed can be accompanied by the formation of toxic oxidation metabolites.
No single test can give a clear picture of the shelf life and oxidative deterioration of a lipid. It is important to understand the characteristics of the specific product and its history in order to run appropriate analytical tests.
Autoxidation mechanism
Lipid autoxidation is the free radical chain reaction between unsaturated lipids and oxygen. The basic mechanism for lipid oxidation consists of three phases.
Initiation. In the presence of initiators, lipids lose a hydrogen radical to form free radicals. The initiators are typically transition metals, ultraviolet light, chemical oxidizers, or enzymes.
Propagation. The free radicals can react with oxygen to form peroxides. These peroxyl radicals further react with lipids to form hydroperoxides to continue the chain reaction mechanism.
Termination. During the last step of autoxidation, the peroxyl radicals interact with each other to form nonradical products. This is where the outward signs of oxidation are manifested with the production of the hydrocarbons, aldehydes, ketones, alcohols, and organic acids associated with rancidity. The most important termination reaction is between antioxidants and peroxyl radicals.
Oxidation factors
Autoxidation is a complex process with many factors that can influence the rate of autoxidative reactions. Although critical to nutritional value, the fatty acid composition of a lipid greatly influences its apparent stability. This is particularly relevant for the polyunsaturated lipids that contain omega-6 and omega-3 fatty acids. For example, docosahexanoic acid has been estimated as up to 250 times more reactive than oleic acid.
Free fatty acid value is another important consideration. Free fatty acids are formed by hydrolytic rancidity caused by the reaction of a lipid and water or by lipase enzymes reacting with lipids. Although the relationship between lipid oxidation and hydrolytic rancidity is not direct, the impacts free fatty acids have on lipid stability are important. It has been reported that free fatty acids react eight to 10 times faster than triglycerides.
In some plant-derived substrates, enzymatic reactions caused by lipoxygenase can also catalyze oxidation of unsaturated fatty acids. This can occur in ingredients such as soybeans and rice bran when they are ground or hydrated. High temperatures or extrusion can inactivate these enzymes, but this should be done soon after grinding or hydration and prior to the enzyme-catalyzed peroxide formation.
Extrusion, moisture effects
The impacts that extrusion can have on product stability shouldn’t be minimized. Products made with high-sheer extrusion that resulted in a higher degree of expansion with larger and thinner cell walls were found more susceptible to oxidation (Hsieh, 1998). This was primarily associated with the increase in product surface area and porosity, which allowed greater oxygen penetration.
Water activity can play an important role in oxidative rancidity. With high water action, oxidation rates increase due to mobilization of free radical initiators such as metal ions. Low water activity can also increase oxidation by removing a protective layer of moisture. Over-drying can cause an increase in oxidation due to the formation of free radicals during drying.
As discussed, metal ions – in particular, iron and copper – can increase rates of oxidation by initiating peroxide formation. A mineral’s chemical form can determine how actively the mineral promotes oxidation. In general, the more water-soluble a mineral is, the more active it will be in promoting oxidation.
Antioxidant options
Antioxidants have been essential components in most animal feeds since the 1960s. They prevent or delay the onset of rancidity during the storage of lipids or foods that contain them.
Antioxidants are generally categorized into two classes: primary and secondary. Primary antioxidants consist of chemical antioxidants that are capable of interrupting the free radical propagation step.
The common synthetic primary antioxidants are butylated hydroxyanisole (BHA), butylated hydroxytoluene (BHT), terbutyl hydroquinone, propyl gallate, and ethoxyquin. All these phenolic compounds can donate hydrogen to free radicals to terminate the oxidation cycle. Secondary antioxidants consist of chemicals that prevent free radical formation and complement the activity of the primary antioxidants. The main secondary antioxidants are metal chelators (citrates and phosphates) and reducing agents (ascorbates and sulfites).
Natural foods trend
Currently there is a growing negative perception of synthetic ingredients, including antioxidants. Several countries have begun to regulate the acceptable amount of residual ethoxyquin in the flesh of fish. Japan has implemented an ethoxyquin limit of less than 1 ppm, and Germany set a limit of 10 ppb. In addition, in 1997, the U.S. Food and Drug Administration requested a voluntary reduction of ethoxyquin in dog food from 150 to 75 ppm.
Consumers are increasingly demanding “natural” human foods and pet foods, resulting in a rise in the organic segments of the market. The move to natural products has many advantages, as the foods have excellent public perception and high consumer awareness, are universally accepted, and are increasingly recognized for their potential health benefits. The primary disadvantages for organic feeds and ingredients are increased costs, higher application rates due to larger molecular weight, and their additional time demands to identify and maintain a unique supply chain. The most common commercially utilized natural antioxidants are tocopherols and rosemary extracts.
Ingredient quality testing
As soon as ingredients or food are manufactured, they are susceptible to oxidative deterioration. That is why it is important to add an antioxidant to feeds as soon as possible. Various analytical methodologies can be utilized to verify quality in both fresh and aged materials. In general, the methods can be categorized according to their purpose: measurement of oxidation byproducts or measurement of resistance to oxidation.
Oxidative byproduct measurements assess the extent of oxidation that has occurred within samples. They commonly test peroxide value, thiobarbituric acid (TBA), anisidine value, and specific aldehydes such as hexanal. Once an antioxidant is depleted within a sample, peroxides begin to accumulate and can be measured. Once the peroxides begin to break down into secondary oxidation byproducts they can be analyzed by TBA, anisidine, or specific aldehyde tests (Fig. 1).
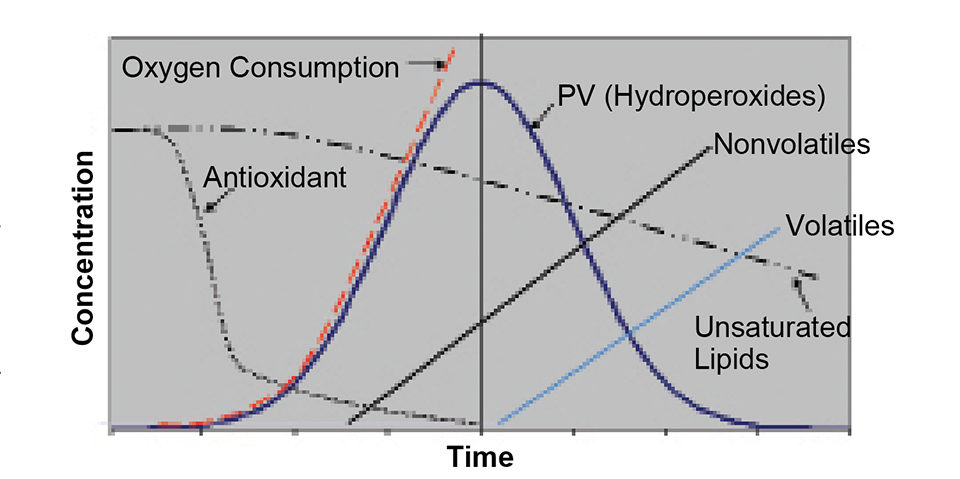
The resistance to oxidation is a measurement of estimated shelf life. The common tests for estimation of stability are the oil stability index, “oxygen bomb,” active oxygen method, and Schall oven test.
Fishmeal testing
Protein meals are extensively used in animal feeds as protein and lipid sources. However, rendered animal byproduct meals are known to be unstable because of their large surface area and unsaturated lipid content. In particular, fishmeal can oxidize rapidly enough that self-heating can occur.
Fishmeal can be tested for protein digestibility, fat percentage and fatty acid profile as indicators of oxidation. Figs. 2 and 3 show the ability of various natural and synthetic antioxidants to protect the omega-3 fatty acids in salmon meal and fish oil. As a measure of raw fish quality, fishmeal can be tested for total volatile nitrogen and biogenic amines (specifically histamine).
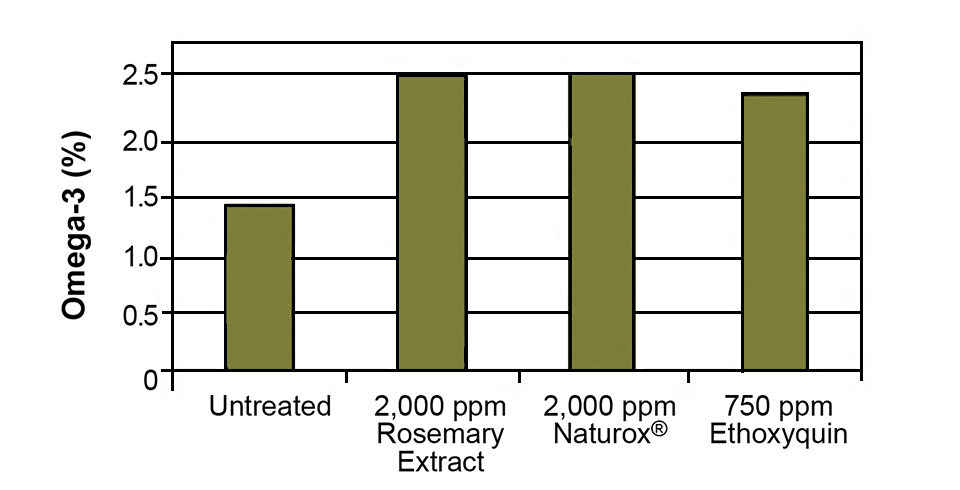
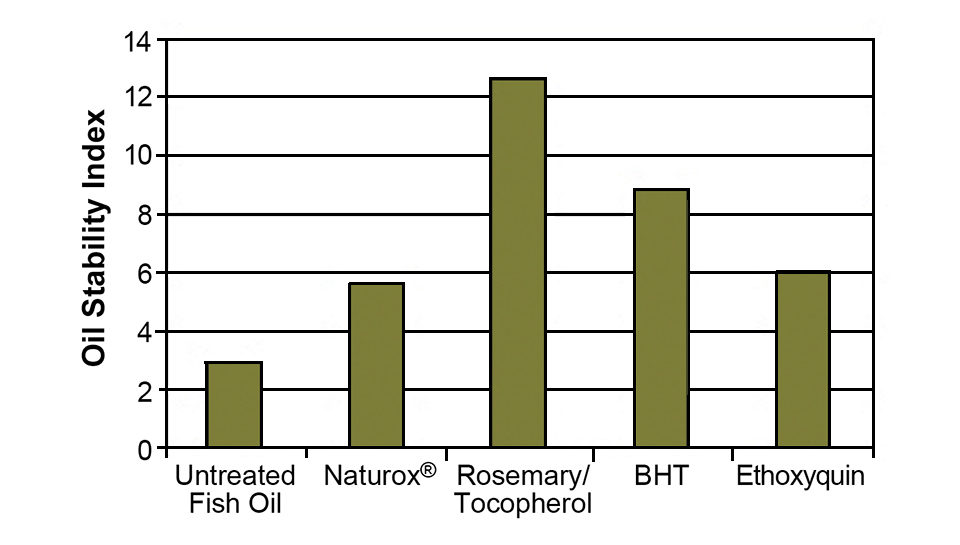
Lipids added to feeds not only add a large portion of the total fat to a diet, but also contribute to diet instability if they are of poor quality or not adequately stabilized with antioxidant. It is valuable to include antioxidants in feed prior to extrusion to help protect the lipids and vitamins from oxidizing during extrusion and drying. However, 50 to 80 percent of the antioxidant content is typically sacrificed through processing. Because of that, the majority of antioxidants are added via surface lipids that also carry the antioxidants to the feed.
Editor’s Note: Cited references are available from the second author.
(Editor’s Note: This article was originally published in the April/May 2006 print edition of the Global Aquaculture Advocate.)
Now that you've reached the end of the article ...
… please consider supporting GSA’s mission to advance responsible seafood practices through education, advocacy and third-party assurances. The Advocate aims to document the evolution of responsible seafood practices and share the expansive knowledge of our vast network of contributors.
By becoming a Global Seafood Alliance member, you’re ensuring that all of the pre-competitive work we do through member benefits, resources and events can continue. Individual membership costs just $50 a year.
Not a GSA member? Join us.
Authors
-

-
Jim Mann
Kemin Nutrisurance
Des Moines, Iowa, USA
Tagged With
Related Posts

Health & Welfare
Ammonia addition enhances microbial flocs in nursery phase for Pacific white shrimp
In a study, “pre-fertilization” in the nursery phase of a biofloc system for shrimp was tested. The objective was to accelerate the biofloc formation to minimize ammonia concentrations, avoiding high peaks during culture.

Responsibility
Ammonia nitrogen dynamics in aquaculture
The major sources of ammonia in aquaculture ponds are fertilizers and feeds, and problems with high ammonia are most common in feed-based aquaculture.

Intelligence
An examination of seafood packaging
Some substances can migrate from plastics and other seafood packaging materials into the product. Even if the substances are not harmful, they can affect the flavor and acceptability of the food.

Aquafeeds
Animal co-product hydrolysates sources of key molecules in aquafeeds
Key molecules found in animal byproduct hydrolysates show potential for use as functional ingredients in aquaculture feeds. Animal co-product hydrolysates from slaughterhouse waste and rendered animal byproducts present a protein alternative.


