Understanding and addressing the enzyme crisis observed in PL stages through optimal ingredient selection and diet formulation can enhance culture performance and overall health

Efficient feed formulation, production, and feeding practices are crucial for the timely delivery of accessible nutrients and their absorption and utilization in shrimp. Yet, at the level of shrimp physiology, available digestive enzymes play a key role in digesting and absorbing proteins that are nutrient-dense molecules. Proteins are essential in the development of larval shrimp diets, providing the necessary amino acids for their growth and survival. Ensuring an adequate protein supply is fundamental to optimizing the health and development of shrimp larvae. Larval molting comes not only with distinct morphological stages but also with profound changes in the digestive system affecting ingestion rate, gut transit time and the enzyme toolbox.
During the early larval stages (zoea-mysis) of marine shrimp, the digestive tract is still very simple, and not fully developed. The early post-larval stage is marked by a significant decrease in the production of digestive enzymes (trypsin and chymotrypsin), a phenomenon referred to as the “enzyme crisis.”
Research indicates that trypsin activity, which drops during the early post-larval stages, significantly declines as the shrimp grows. Similarly, chymotrypsin activity increases during the zoea stages but drops during the early post-larval stages. This reduction in enzyme production challenges the shrimp’s ability to efficiently digest and utilize dietary proteins during this critical period of growth and development. Understanding this enzyme crisis is essential for formulating effective diets that support optimal growth in shrimp aquaculture.
Our study, conducted in collaboration with the laboratory of Prof. Dr. Daniel Lemos from the University of Sao Paulo (Brazil), focused on optimizing the in vitro digestibility assay for larval and post-larval stages of Pacific white shrimp (Litopenaeus vannamei). We also conducted a comprehensive screening of various ingredients to analyze their digestibility, understand the impact of the enzyme crisis, and identify ways to support optimal growth during these stages.
Materials and methods
We applied the pH-stat assay as an in vitro method to evaluate the protein digestibility of various ingredients, including marine and terrestrial plant and animal-derived protein sources.
The pH-stat method for in vitro protein digestibility was standardized using enzyme extracts from different zoea (Z), mysis (M), and post-larval (PL) stages of L. vannamei. The optimal enzyme-to-substrate ratio (E:S) was determined by evaluating different concentrations to achieve repeatable values and significant protein hydrolysis.

This method involves monitoring protein hydrolysis through changes in pH. During proteolysis, protons are released from cleaved peptide bonds at an alkaline pH and cause a decrease in pH. This reduction is countered by continuously adding sodium hydroxide (NaOH) to maintain a constant pH, allowing the measurement of NaOH consumption, which correlates with the degree of hydrolysis and, consequently, protein digestibility. The pH-stat method is considered reliable due to its simplicity and precision when assessing the digestibility of various feed ingredients.
Apparent digestibility of six novel protein sources in Pacific white shrimp diets
Results and discussion
Digestibility by stage
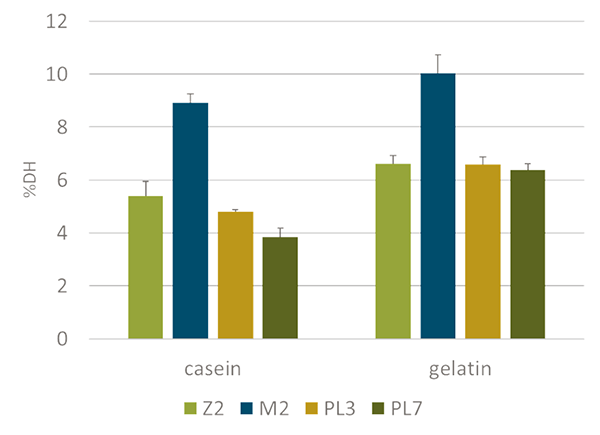
The assay revealed significant variations in protein digestibility across different developmental stages of L. vannamei and underscored the importance of stage-specific dietary formulations to optimize shrimp growth and health. Specifically, the mysis II (MII) stage exhibited higher protein digestibility compared to other stages. The data indicated an increase in enzymatic potential to digest proteins from the zoea ZII stage to MII, followed by a decrease from MII to postlarva PL3, and an ongoing decrease from PL3 to PL7 (Fig. 1).
Digestibility of different ingredients
The selection of ingredients is crucial for formulating effective larval diets. Our study examined the digestibility of various feed ingredients across different developmental stages. We utilized two controls (casein and gelatin), five marine sources, two plant sources, and one yeast source (Fig. 2). The highest degree of hydrolysis was observed for freeze-dried fish, followed by squid meal and gelatin.
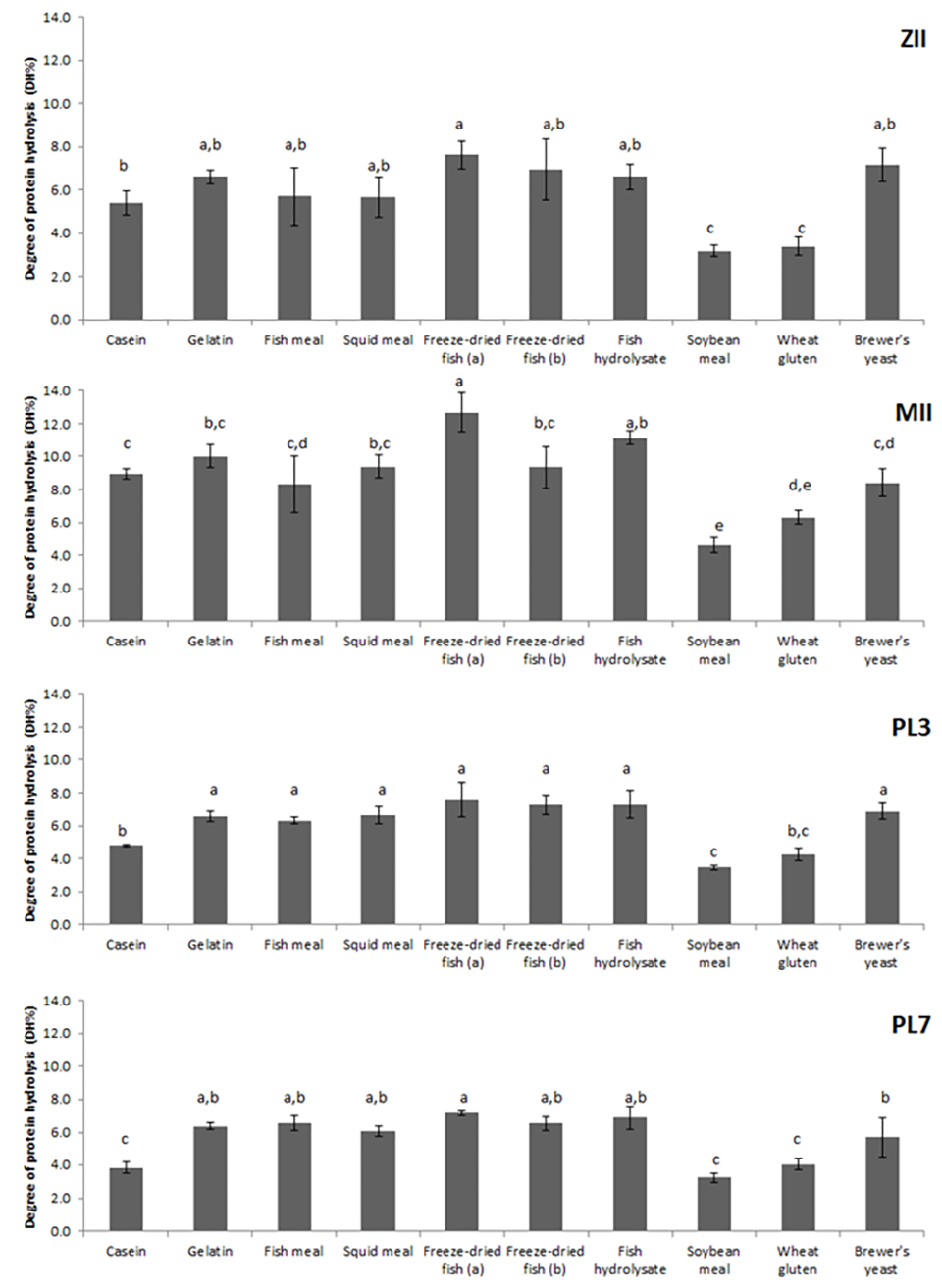
The marine protein sources were generally more digestible than plant sources. This disparity can be attributed to the presence of enzyme inhibitors in plant sources and the lack of specific proteases required for the efficient digestion of plant material by shrimp larvae. For instance, soybean meal, a common plant protein, exhibited lower digestibility across all stages in our study.
Freeze-dried fish was the most digestible ingredient in all stages, highlighting the superior nutritional value of marine-based feeds. The relatively lower degree of hydrolysis with fish meal compared to freeze-dried fish further illustrates how raw material processing can affect digestibility. Proteins from fresh-frozen seafood, for example, are considered high-quality sources due to their enhanced digestibility and nutritional content. These fresh-frozen marine ingredients provide essential nutrients that are readily absorbed by the larvae, supporting their growth and development more effectively than processed dry marine ingredients.
The enzyme crisis observed in the post-larval stages PL3 and, to a lesser extent PL7, poses a significant challenge to protein digestion and nutrient absorption. Understanding and addressing this enzyme crisis through optimal ingredient selection and diet formulation can enhance culture performance and overall health.
Conclusions
First, the screening of different ingredients demonstrated that marine sources had consistently higher digestibility compared to plant sources for the stages investigated. Second, the higher digestibility of freeze-dried fish compared to fish meal showed how increased ingredient processing affects digestibility and highlighted the potential of fresh-frozen ingredients. Third, the pH-stat method proved an effective model for determining the protein digestibility of different feed ingredients.
Now that you've reached the end of the article ...
… please consider supporting GSA’s mission to advance responsible seafood practices through education, advocacy and third-party assurances. The Advocate aims to document the evolution of responsible seafood practices and share the expansive knowledge of our vast network of contributors.
By becoming a Global Seafood Alliance member, you’re ensuring that all of the pre-competitive work we do through member benefits, resources and events can continue. Individual membership costs just $50 a year.
Not a GSA member? Join us.
Authors
-
Celine De Maesschalck, Ph.D.
Corresponding author
INVE Technologies NV, Hoogveld 93, 9200 Dendermonde, Belgium -
Roeland Wouters, Ph.D.
INVE Technologies NV, Hoogveld 93, 9200 Dendermonde, Belgium
-
Olivier Decamp, Ph.D.
INVE (THAILAND) Ltd, Amphoe Wachirabarami, Phichit 66220, Thailand
-
Eva Werbrouck, Ph.D.
INVE Technologies NV, Hoogveld 93, 9200 Dendermonde, Belgium
Tagged With
Related Posts

Aquafeeds
Animal protein meals reduce feed costs but don’t improve shrimp performance
The authors conducted a study to determine how replacement of salmon meal with various animal protein meals in feed affected the growth performance of white shrimp.

Health & Welfare
Determining causes of osmotic stress in Pacific white shrimp postlarvae
This study evaluated the relationship between the molting stage, age and osmotic stress in Pacific white shrimp postlarvae. The results indicate that osmotic stress tests should be done only with at least PL12 at the intermolt stage and that were cultured at 35 ppt salinity and 29 degrees-C.

Health & Welfare
Natural strategies to improve growth and health of farmed shrimp
The application of probiotics, prebiotics and lipopolysaccharides has shown positive results with farmed shrimp, and is a very promising field, but more research is needed to clarify and foresee the molecular impact of these biological entities.

Aquafeeds
The digestibility of hydrolyzed soy protein
Studies demonstrate that hydrolyzed soy protein can be well digested and utilized by Pacific white shrimp, and that digestibility is improved through a bioprocess with the bacterium Lactobacillus spp.



