Optimization of host animals’ gut microbial flora leads to better growth, survival

The production of marine fish juveniles in commercial hatcheries largely depends on live preys such as rotifers and artemia as food sources. Optimization of the zootechnical, nutritional and microbial factors of live foods can reduce the commonly heavy mortalities of the fish larvae.
Although live preys are recognized as one of the pathways for the entry of pathogenic bacteria into fish larvae, they can also function as possible vectors for the delivery of nutrients, antimicrobial agents, vaccines and probiotics to larval fish. For example, research with varied fish species has shown that Artemia urmiana nauplii can be bioencapsulated with beneficial microorganisms.
Persian sturgeon larvae

In research, using A. urmiana nauplii in the process of bioencapsulation or enrichment by probiotic bacilii showed the artemia had a high potential to carry the produced suspension of bacteria. The high grazing rate of A. urmiana nauplii on bacteria illustrated that it can be used as a suitable vector to carry probiotic bacteria into the digestive tracts of cultured fish.
The probiotic Bacillus species were successfully incorporated into the A. urmiana nauplii at a level of 5.67 x 102 colony-forming units (CFU)/nauplii during a period of 10 hours. The concentration of bacteria in broth was 1 x 105 CFU/ml.
When larvae of Persian sturgeon (Acipenser persicus) were fed bioencapsulated A. urmiana nauplii at 30 percent of their body weight daily, a high concentration (6 x 107 CFU/larvae) of probiotic Bacillus was counted in the digestive tracts of the fish after 30 days of feeding.
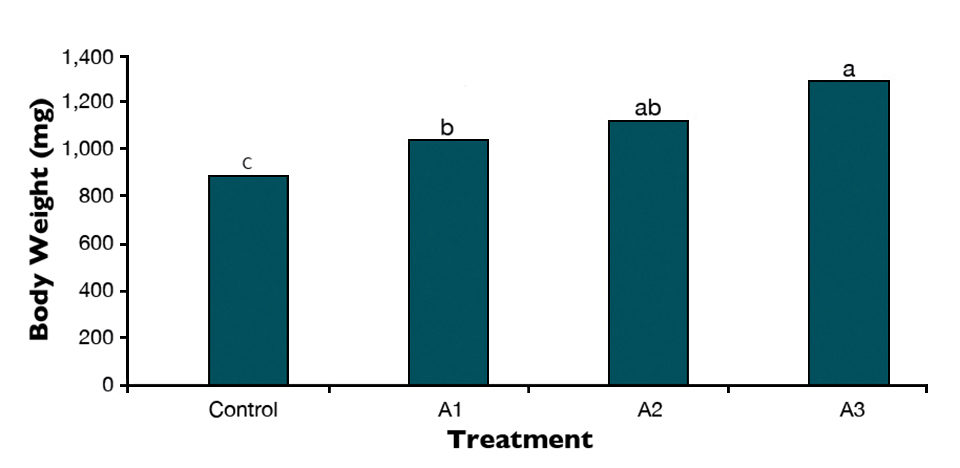
This resulted in a survival rate of 89 percent, feed efficiency of 43.96 percent, total amylase of 1.8 international units (I.U.)/mg protein, protease of 0.37 I.U./mg protein and lipase of 1.66 I.U./mg protein. The related values for the control were 79 percent, 31.99 percent, 0.87 I.U./mg protein, 0.19 I.U./mg protein and 1.01 I.U./mg protein. The body weight of the sturgeon larvae increased from 901 mg for the control to 1,233 mg (Figure 1). Also, the survival time for Persian sturgeon larvae challenged with salinity stress increased significantly.
Spiny sturgeon larvae
In another study, inistar-I A. urmiana nauplii were fed to larval-stage spiny sturgeons, Acipenser nudiventris, as a vector to carry probiotics to the larvae’s digestive tracts. The Artemia were bioencapsulated at different levels of bacterial suspensions for 10 hours. The sturgeon larvae were fed 30 percent of their body weight with enriched A. urmiana nauplii six times a day.
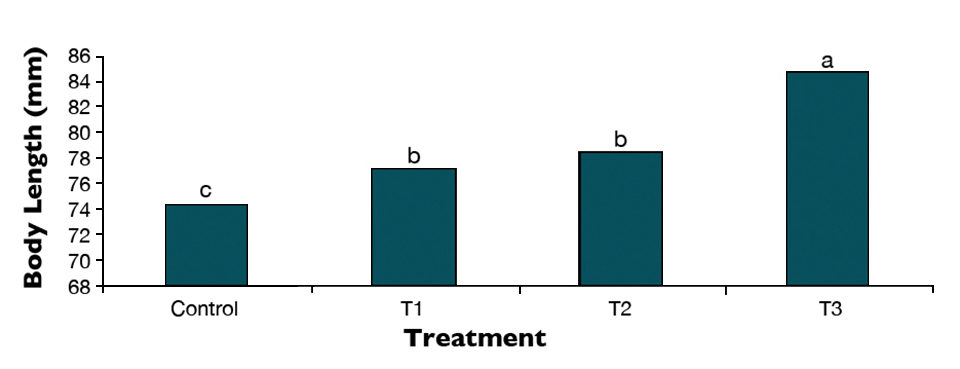
The results indicated that the A. urmiana nauplii microenriched by the Bacillus significantly increased final body weights, final body lengths (Fig. 2) and specific growth rates in the experimental fish in comparison with the control. The study also showed that different concentrations of probiotics in the bioencapsulation broth of A. urmiana nauplii could have different effects on growth performance and feeding efficiency of the sturgeon larvae.
Beluga sturgeon larvae
Newly hatched A. urmiana nauplii were also employed for early feeding of larval beluga sturgeon, Huso huso. In this study, in which the Artemia nauplii carried the probiotic Bacillus to the digestive tracts of the larvae, results showed that the carcass proximate compositions of the larvae were significantly increased. The enriched artemia nauplii improved food intake, protein productive value, nitrogen retained efficiency, protein gain and energy retained as protein.
Silver carp larvae
Other research with larval silver carp, Hypophthalmichthys molitrix, showed that A. urmiana nauplii grazed the spores of profitable bacteria and had a high rate of inoculation with them. Spores of Bacillus circulans and B. lichenifirmis were used in the bioencapsulation suspension broth. Significant positive results for conversion efficiency ratio, condition factor, daily growth, specific growth rate and feed-conversion efficiency were obtained in the fish fed probiotics via bioencapsulation of A. urmiana nauplii.
Koi carp larvae
A. urmiana nauplii were also used as a vector to deliver the bakers yeast Saccharomyces cerevisiae into the digestive tracts of larval koi carp, Cyprinus carpio carpio. Fish treated with the yeast and probiotic Bacillus showed positive responses in such growth parameters as weight gain, feed-conversion ratio and specific growth. Significant positive correlations were observed between the concentration of probiotics in bacterial broth suspension of the artemia nauplii and weight gain (r = 0.723, p < 0.05), specific growth (r = 0.669, p < 0.05) and conversion efficiency ratio (r = 0.776, p < 0.01) of the carp larvae.
The bioencapsulation resulted in higher culture performance, with the best results in the trial in which fish larvae were fed artemia nauplii with 3 x 108 CFU yeast and Bacillus/L.
Perspectives
A. urmiana nauplii enriched with useful microorganisms can help in the optimization and development of gut microbial flora of host animals and lead to increased growth parameters and survival. Through such improvements, bioencapsulation can decrease the overall cost of aquaculture production and become a significant factor in the development of the industry.
(Editor’s Note: This article was originally published in the November/December 2010 print edition of the Global Aquaculture Advocate.)
Now that you've reached the end of the article ...
… please consider supporting GSA’s mission to advance responsible seafood practices through education, advocacy and third-party assurances. The Advocate aims to document the evolution of responsible seafood practices and share the expansive knowledge of our vast network of contributors.
By becoming a Global Seafood Alliance member, you’re ensuring that all of the pre-competitive work we do through member benefits, resources and events can continue. Individual membership costs just $50 a year.
Not a GSA member? Join us.
Author
-
Hojatolah Jafaryan, Ph.D.
Department of Fisheries
Gonbad Institute of Higher Education
Golestan Province, Iran
Tagged With
Related Posts

Health & Welfare
Assessment of supplemental Bacillus probiotics in whiteleg shrimp juveniles
Results of a feeding study supplementing probiotics in the diet showed that when the Bacillus species were complemented in an appropriate concentration into feeds, the growth and feed efficiency of whiteleg shrimp could be improved.

Health & Welfare
Bacillus probiotics benefit tilapia rearing under challenging conditions in Brazil
A recent study that evaluated the benefits of using probiotics with a balanced mixture of Bacillus bacteria strains to inhibit pathogenic bacteria in tilapia.

Health & Welfare
Bacillus probiotics improve hatchery, nursery production in EMS-hit Mexico
In early 2014, a trial to evaluate the effects of a mixture of Bacillus strains on early mortality syndrome bacteria during the larviculture and nursery phases for shrimp was carried out at a commercial hatchery in Mexico.

Health & Welfare
Culture water pre-inoculation of probiotics in Pacific white shrimp nurseries
This study showed that a longer inoculation of probiotics will improve shrimp postlarvae growth and survival, and that a new technique developed for more accurately estimating postlarvae weight can significantly reduce the food that must be supplied.



